- Polytech
EMNS
EMNS - Engineering of Molecular NanoSystems
WELCOME !
The EMNS Lab is part of the Chemistry and Materials Science Department of the Brussels School of Engineering and is active in various research topics with applications mainly in the field of Health and Environment.
NANOMATERIALS FOR SENSING APPLICATIONS
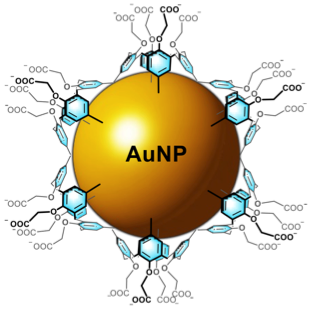
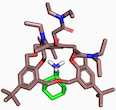
MOLECULAR RECOGNITION: from organic solvents to water
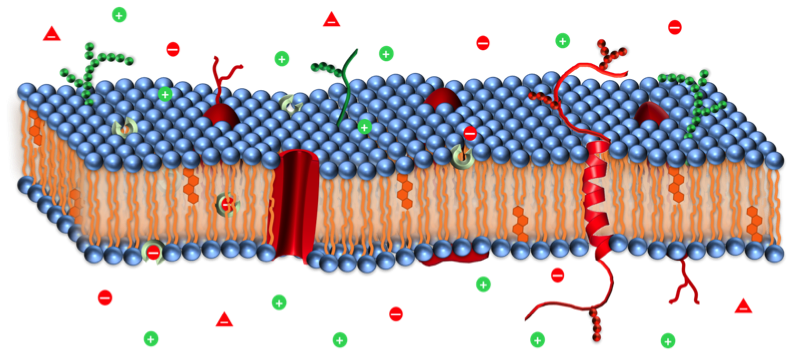
ION TRANSPORT ACROSS LIPID BILAYERS
1st LUCES Global Action Meeting in Abano Terme
19 avril 2024
The EMNS lab is in the core of the new COST Action "Supramolecular LUminescent Chemosensors for Environmental Security" (LUCES).
New publication in ACS Applied Nano Materials
8 mars 2024
Tailored Ultrastable Core–Shell Au@Ag Nanoparticles for Enhanced Colorimetric Detection in Lateral Flow Assays, by Bryan Gosselin, Gilles Bruylants and Ivan Jabin - First published: 05 March 2024
EMNS researchers go abroad for advanced experiments
20 février 2024
A short tour of Europe this February...
LUCES COST Action's Core Group meets in Lisbon
20 février 2024
Kristin and Mélanie, respectively Vice-Chair and Grant Holder Manager, are in the core of the new COST Action "Supramolecular Luminescent Chemosensors for Environmental Security" (LUCES).
New publication in Angewandte Chemie
12 janvier 2024
Tuning CH Hydrogen Bond-Based Receptors toward Picomolar Anion Affinity via the Inductive Effect of Distant Substituents, by Matúš Chvojka, Dr. Dominik Madea, Dr. Hennie Valkenier, Prof. Vladimír Šindelář - First published: 08 December 2023
